23239-88-5
- Product Name:Benzocaine hydrochloride
- Molecular Formula:C9H11NO2.HCl
- Purity:99%
- Molecular Weight:201.653
Product Details;
CasNo: 23239-88-5
Molecular Formula: C9H11NO2.HCl
Appearance: hite crystalline powder
Hot Sale Fast Delivery Benzocaine hydrochloride 23239-88-5 In Medicine
- Molecular Formula:C9H11NO2.HCl
- Molecular Weight:201.653
- Appearance/Colour:hite crystalline powder
- Vapor Pressure:0.000589mmHg at 25°C
- Melting Point:208oC
- Boiling Point:310.7 °C at 760 mmHg
- Flash Point:164.2 °C
- PSA:52.32000
- Density:1.286g/cm3
- LogP:2.82870
Benzocaine hydrochloride(Cas 23239-88-5) Usage
|
Description |
Benzocaine is a local anesthetic drug. It is directly applied to the skin and used as a topical pain reliever or in cough drops. Benzocaine works by creating a chemical barrier that halts the build up of sodium, which accumulates as the nerve endings are stimulated by pain. When sodium builds up, electrical signals also build in the nerve ending and are transmitted to the brain, which interprets the signals as pain. Benzocaine hydrochloride is a salt modification of benzocaine, formed when benzocaine is complexed with hydrochloric acid. Compared to benzocaine, benzocaine hydrochloride is more water soluble, making it more appropriate for oral administration. Benzocaine hydrochloride is usually made into powder or oil paste and used to heal wounds, ulcers, burns, skin abrasion, and hemorrhoids. Through the reduction in the excretion of ammonia and carbon dioxide of fish by benzocaine hydrochloride, the pH and alkalinity values of the transport water remains constant, thereby, benzocaine hydrochloride is used as an aid in the transport of fish. |
|
Uses |
Benzocaine Hydrochloride can be used as promising MAO inhibitors. |
InChI:InChI=1/C9H11NO2.ClH/c1-2-12-9(11)7-3-5-8(10)6-4-7;/h3-6H,2,10H2,1H3;1H
23239-88-5 Relevant articles
Cyclometalated Ruthenium(II) NHC Complexes with Imidazo[1,5-a]pyridine-Based (C^C*) Ligands – Synthesis and Characterization
Schleicher, David,Tronnier, Alexander,Soellner, Johannes,Strassner, Thomas
, p. 1956 - 1965 (2019/03/07)
We present the synthesis and characteriz...
Organosilicon synthesis of isocyanates: III. Synthesis of aliphatic, carbocyclic, aromatic, and alkylaromatic isocyanatocatboxylic acid esters
Lebedev,Lebedeva,Sheludyakov,Ovcharuk,Kovaleva,Ustinova
, p. 1069 - 1080 (2008/02/05)
A series of aminoacid esters was prepare...
AMINOPHENYLPROPANOIC ACID DERIVATIVE
-
Page/Page column 120, (2010/11/24)
A compound represented by the formula (1...
Hydrogen bond controlled aggregation of guanidinium-carboxylate derivatives in the solid state
Zafar, Abdullah,Melendez, Rosa,Geib, Steven J,Hamilton, Andrew D
, p. 683 - 690 (2007/10/03)
In this paper, we report the synthesis a...
23239-88-5 Process route
-
-
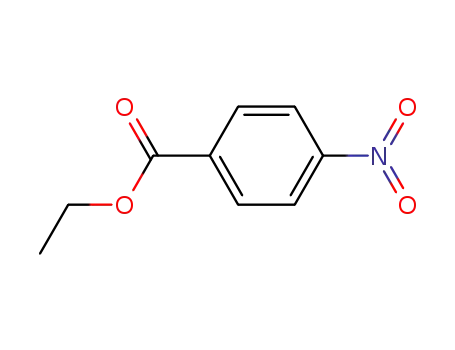
99-77-4
ethyl 4-nitrobenzoate

-
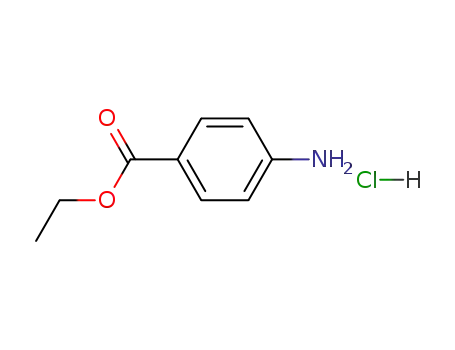
-
23239-88-5
benzocaine hydrochloride
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride; hydrogen;
palladium on activated charcoal;
In
ethanol;
for 0.5h;
under 2585.7 Torr;
|
-
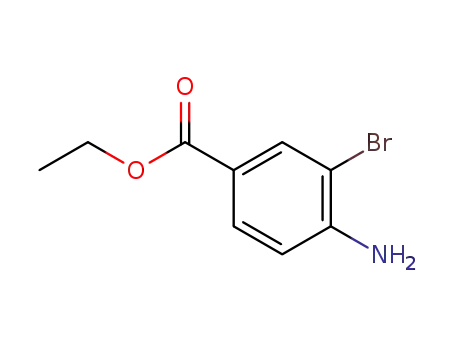
-
7149-03-3
ethyl 3-bromo-4-aminobenzoate

-
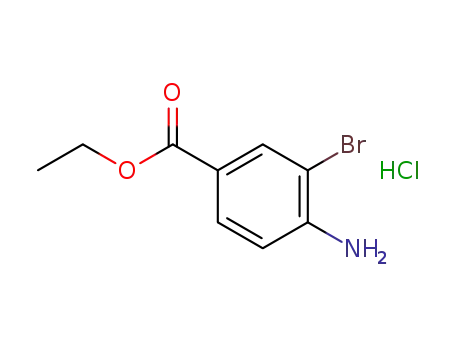
-
865139-46-4
ethyl 4-amino-3-bromobenzoate hydrochloride

-

-
23239-88-5
benzocaine hydrochloride
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
ethyl acetate;
|
23239-88-5 Upstream products
-
99-77-4

ethyl 4-nitrobenzoate
-
64-17-5
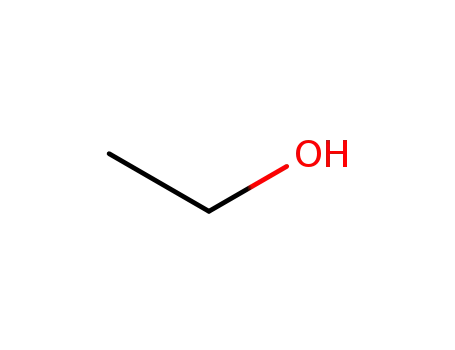
ethanol
-
150-13-0
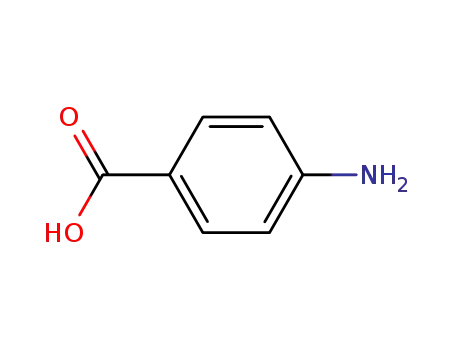
4-amino-benzoic acid
-
7149-03-3

ethyl 3-bromo-4-aminobenzoate
23239-88-5 Downstream products
-
30806-83-8
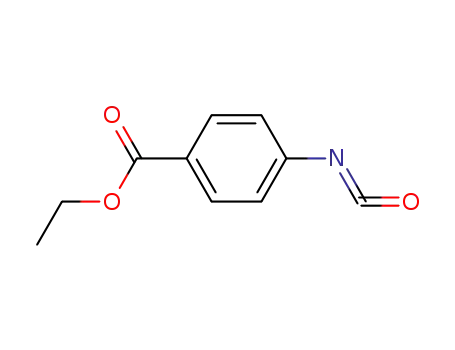
p-ethoxycarbonylphenyl isocyanate
-
102660-78-6
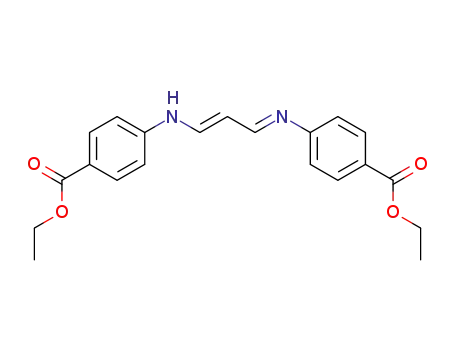
3-(4-ethoxycarbonyl-anilino)-acrylaldehyde-(4-ethoxycarbonyl-phenylimine)
-
116132-26-4
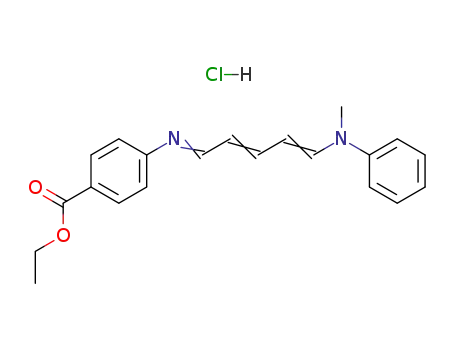
4-[5-(N-methyl-anilino)-penta-2,4-dienylidenamino]-benzoic acid ethyl ester; hydrochloride
-
102892-96-6
ethyl 1-p-benzoate-2,5-di-p-bromopyrrole
Relevant Products
-
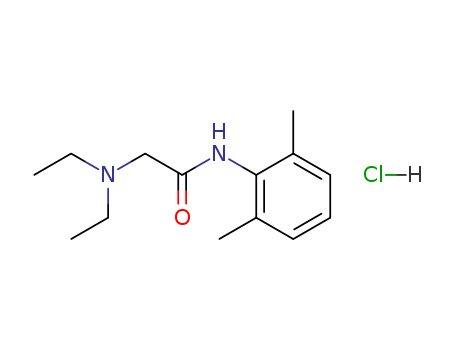
Lidocaine hydrochloride
CAS:73-78-9
-
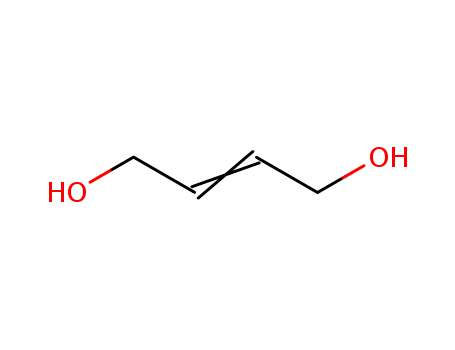
2-Butene-1,4-diol
CAS:110-64-5
-
2-Benzylamino-2-methyl-1-propanol
CAS:10250-27-8