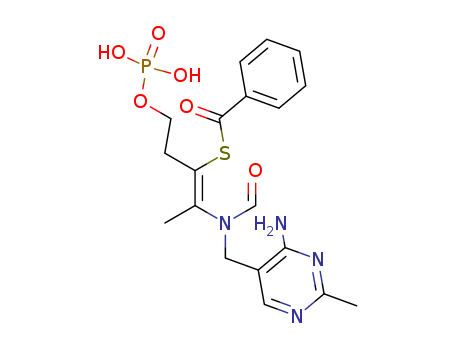
159752-10-0
- Product Name:MK-677
- Molecular Formula:C28H40N4O8S2
- Purity:99%
- Molecular Weight:624.78
Product Details;
CasNo: 159752-10-0
Molecular Formula: C28H40N4O8S2
Best Price Sells MK-677 159752-10-0 In Stock
- Molecular Formula:CH4O3S*C27H36N4O5S
- Molecular Weight:624.78
- Boiling Point:868.9 °C at 760 mmHg
- Flash Point:479.3 °C
- PSA:196.66000
- LogP:4.97440
159752-10-0 Relevant articles
Domino Aryne Annulation via a Nucleophilic-Ene Process
Xu, Hai,He, Jia,Shi, Jiarong,Tan, Liang,Qiu, Dachuan,Luo, Xiaohua,Li, Yang
, p. 3555 - 3559 (2018/03/21)
1,2-Benzdiyne equivalents possess the un...
A facile synthesis of the spiroindoline-based growth hormone secretagogue, MK-677
Qi, Xian Liang,Yang, Er Qun,Zhang, Jun Tao,Wang, Tao,Cao, Xiao Ping
, p. 661 - 664 (2012/07/03)
A facile and improved route for the synt...
Polymorphic forms of a growth hormone secretagogue
-
, (2008/06/13)
This invention is concerned with polymor...
Synthesis of the orally active spiroindoline-based Growth Hormone Secretagogue, MK-677
Maligres, Peter E.,Houpis, Ioannis,Rossen, Kai,Molina, Audrey,Sager, Jess,Upadhyay, Veena,Wells, Kenneth M.,Reamer, Robert A.,Lynch, Joseph E.,Askin, David,Volante,Reider, Paul J.,Houghton, Peter
, p. 10983 - 10992 (2007/10/03)
The preparation of the Merck Growth Horm...
159752-10-0 Process route
-
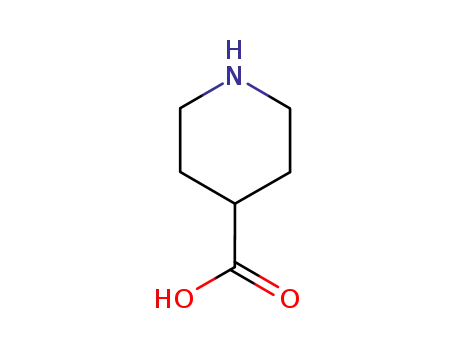
-
498-94-2
isonipecotic acid

-
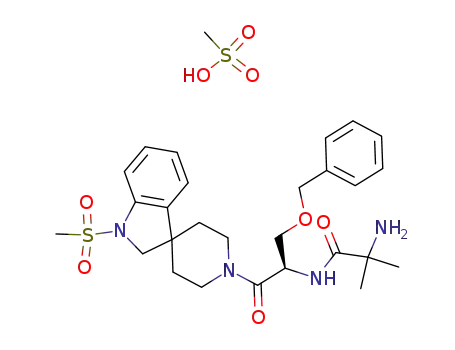
-
191487-52-2,214962-40-0,159752-10-0
MK-677
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 12 steps
1: 97 percent / K2CO3 / H2O / 58 h / 22 °C
2: (COCl)2 / toluene; dimethylformamide / 16 h / 18 °C
3: 94 percent / H2, DIEA, thioanisole / Pd/C / toluene / 22 h / 20 °C / 2068.6 Torr
4: 99 percent / TFA / CH2Cl2 / 17 h / 35 °C
5: NaBH4 / toluene / 0.5 h / -2 °C
6: DIEA / tetrahydrofuran / 2 h / 5 - 8 °C
7: 93 percent / H2 / Pd-C / ethanol / 5 h / 65 °C / 2068.6 Torr
8: DCC, HOBt / H2O; various solvent(s) / 5 h / Ambient temperature
9: MsOH / ethanol / 7.5 h / 35 - 40 °C
10: DCC, HOBt / various solvent(s) / 2 h / Ambient temperature
11: MsOH / ethanol / 35 - 40 °C
12: ethanol; ethyl acetate / 55 °C
With
sodium tetrahydroborate; oxalyl dichloride; methanesulfonic acid; methyl-phenyl-thioether; hydrogen; potassium carbonate; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine; dicyclohexyl-carbodiimide; trifluoroacetic acid;
palladium on activated charcoal;
In
tetrahydrofuran; ethanol; dichloromethane; water; ethyl acetate; N,N-dimethyl-formamide; toluene;
|
|
|
Multi-step reaction with 10 steps
1: lithium aluminium tetrahydride / tetrahydrofuran / 20 °C
2: potassium carbonate / 20 °C
3: pyridinium chlorochromate / dichloromethane / 20 °C
4: trifluoroacetic acid / dichloromethane
5: sodium tetrahydroborate / methanol
6: N-ethyl-N,N-diisopropylamine / tetrahydrofuran
7: palladium 10% on activated carbon; hydrogen / ethanol
8: benzotriazol-1-ol; dicyclohexyl-carbodiimide / Isopropyl acetate; water
9: benzotriazol-1-ol; dicyclohexyl-carbodiimide / Isopropyl acetate; water
10: ethanol
With
sodium tetrahydroborate; lithium aluminium tetrahydride; palladium 10% on activated carbon; hydrogen; potassium carbonate; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine; dicyclohexyl-carbodiimide; pyridinium chlorochromate; trifluoroacetic acid;
In
tetrahydrofuran; methanol; ethanol; dichloromethane; Isopropyl acetate; water;
|
-
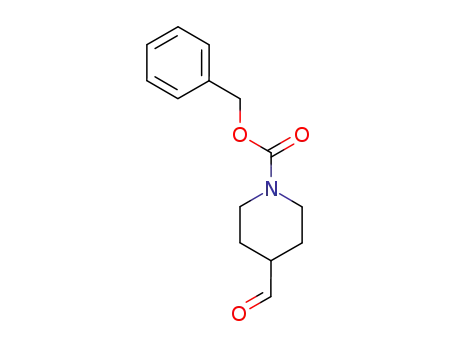
-
138163-08-3
N-(benzyloxycarbonyl)piperidine-4-carboxaldehyde

-

-
191487-52-2,214962-40-0,159752-10-0
MK-677
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 9 steps
1: 99 percent / TFA / CH2Cl2 / 17 h / 35 °C
2: NaBH4 / toluene / 0.5 h / -2 °C
3: DIEA / tetrahydrofuran / 2 h / 5 - 8 °C
4: 93 percent / H2 / Pd-C / ethanol / 5 h / 65 °C / 2068.6 Torr
5: DCC, HOBt / H2O; various solvent(s) / 5 h / Ambient temperature
6: MsOH / ethanol / 7.5 h / 35 - 40 °C
7: DCC, HOBt / various solvent(s) / 2 h / Ambient temperature
8: MsOH / ethanol / 35 - 40 °C
9: ethanol; ethyl acetate / 55 °C
With
sodium tetrahydroborate; methanesulfonic acid; hydrogen; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine; dicyclohexyl-carbodiimide; trifluoroacetic acid;
palladium on activated charcoal;
In
tetrahydrofuran; ethanol; dichloromethane; water; ethyl acetate; toluene;
|
|
|
Multi-step reaction with 7 steps
1: trifluoroacetic acid / dichloromethane
2: sodium tetrahydroborate / methanol
3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran
4: palladium 10% on activated carbon; hydrogen / ethanol
5: benzotriazol-1-ol; dicyclohexyl-carbodiimide / Isopropyl acetate; water
6: benzotriazol-1-ol; dicyclohexyl-carbodiimide / Isopropyl acetate; water
7: ethanol
With
sodium tetrahydroborate; palladium 10% on activated carbon; hydrogen; benzotriazol-1-ol; N-ethyl-N,N-diisopropylamine; dicyclohexyl-carbodiimide; trifluoroacetic acid;
In
tetrahydrofuran; methanol; ethanol; dichloromethane; Isopropyl acetate; water;
|
159752-10-0 Upstream products
-
498-94-2

isonipecotic acid
-
47173-80-8
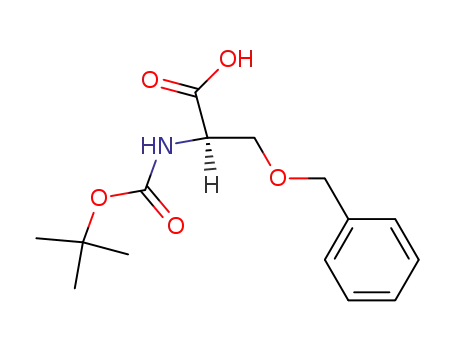
N-Boc-D-serine(Bzl)-OH
-
100-39-0
benzyl bromide
-
10314-98-4
1-benzyloxycarbonylpiperidine-4-carboxylic acid
Relevant Products
-
Benfotiamine
CAS:22457-89-2
-
N,N-Diethyl-3-methylbenzamide
CAS:134-62-3
-
2-Methoxyethyl ether
CAS:111-96-6