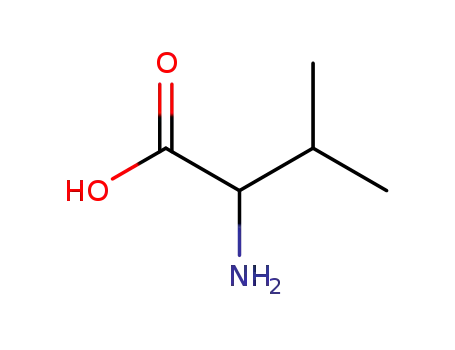
72-18-4
- Product Name:L-Valine
- Molecular Formula:C5H11NO2
- Purity:99%
- Molecular Weight:117.148
Product Details;
CasNo: 72-18-4
Molecular Formula: C5H11NO2
Appearance: White crystalline powder
Good Supplier In China Fast Delivery L-Valine 72-18-4 In Bulk Supply
- Molecular Formula:C5H11NO2
- Molecular Weight:117.148
- Appearance/Colour:White crystalline powder
- Melting Point:295-300 °C (subl.)(lit.)
- Refractive Index:28 ° (C=8, HCl)
- Boiling Point:213.642 °C at 760 mmHg
- PKA:2.37±0.10(Predicted)
- Flash Point:83.008 °C
- PSA:63.32000
- Density:1.064 g/cm3
- LogP:0.75460
- IDLH:1426
- IDLH:3444
L-Valine(Cas 72-18-4) Usage
| Uses | L-Valine is one of the twenty common amino acids that serve as the building blocks of proteins. It is classified as a branched-chain essential amino acid (BCAA), which means it cannot be synthesized by the human body and must be obtained through the diet. |
|
Who Evaluation |
Evaluation year: 2004 |
InChI:InChI:1S/C5H11NO2/c1-3(2)4(6)5(7)8/h3-4H,6H2,1-2H3,(H,7,8)
72-18-4 Relevant articles
Engineering of microbial cells for L-valine production: challenges and opportunities
Hui Gao, Philibert Tuyishime, Xian Zhang, Taowei Yang, Meijuan Xu & Zhiming Rao
, Microbial Cell Factories volume 20, Article number: 172 (2021)
The sites responsible for L-valine feedback inhibition are located in the small subunits of AHAS I (encoded by ilvN) and AHAS III (encoded by ilvH), while AHAS II (encoded by ilvGM) is resistant to L-valine. When L-valine is abundant in cells, CodY will inhibit ilvBHC, ilvA, ilvD, ygaE, ywaA. L-valine is decomposed to 2-ketoisovalerate through the enzyme Bcd which is also known as leucine dehydrogenase (LeuDH).
Cyclic Tetrapeptides with Synergistic Antifungal Activity from the Fungus Aspergillus westerdijkiae Using LC-MS/MS-Based Molecular Networking
Chen, Baosong,Dai, Huanqin,Han, Junjie,Li, Erwei,Liu, Hongwei,Lyu, Zhitang,Song, Fuhang,Sun, Jingzu,Wang, Hanying,Wang, Tao,Wang, Wenzhao,Zhang, Rui
, (2022/02/17)
Fungal natural products play a prominent...
High-yield production of L-valine in engineered Escherichia coli by a novel two-stage fermentation
Yanan Hao a 1, Qian Ma a b 1, Xiaoqian Liu a, Xiaoguang Fan a b, Jiaxuan Men a, Heyun Wu a, Shuai Jiang a, Daoguang Tian a, Bo Xiong a, Xixian Xie a b
, Metabolic Engineering Volume 62, November 2020, Pages 198-206
For efficient efflux of L-valine, an exporter from Corynebacterium glutamicum was subsequently introduced. Next, the precursor pyruvate pool was increased by knockout of GTP pyrophosphokinase and introduction of a ppGpp 3′-pyrophosphohydrolase mutant to facilitate the glucose uptake process.
72-18-4 Upstream products
-
516-06-3
D,L-valine
-
4289-97-8
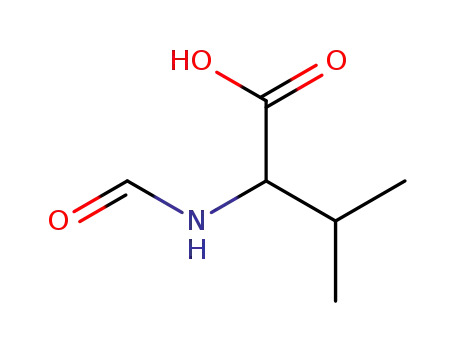
N-formylvaline
-
2901-80-6
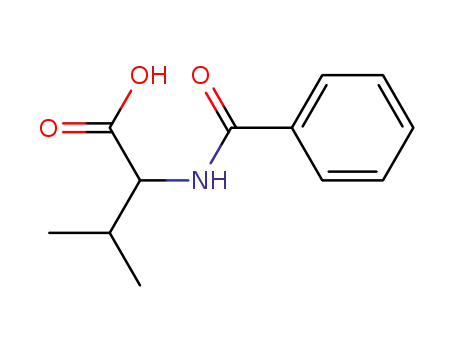
N-benzoyl-D,L-valine
-
25692-88-0
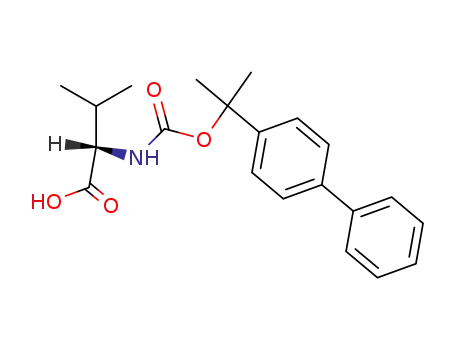
Nα-[2-(4-biphenylyl)-2-propyloxycarbonyl]-L-valine
72-18-4 Downstream products
-
6306-52-1
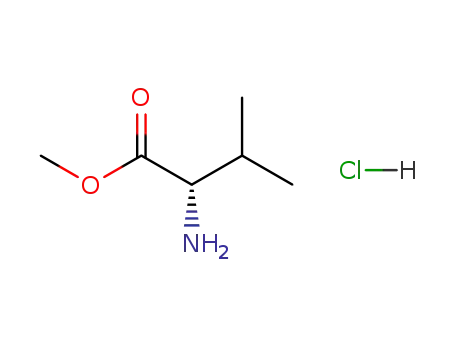
L-valine methylester hydrochloride
-
6306-54-3
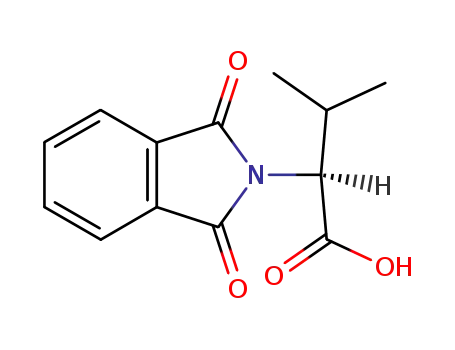
N-phthaloyl L-valine
-
10003-64-2
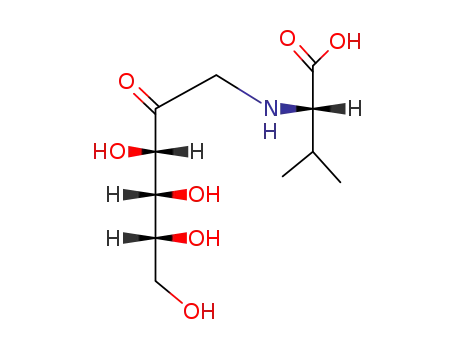
N-(1-deoxy-D-fructos-1-yl)-L-valine
-
349-00-8
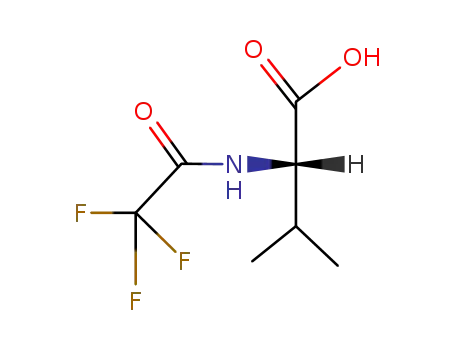
N-trifluoroacetyl-L-valine
Relevant Products
-
Tropinone
CAS:532-24-1
-
2-Methoxyethyl ether
CAS:111-96-6
-
4-Amino-3-phenylbutanoic acid
CAS:1078-21-3