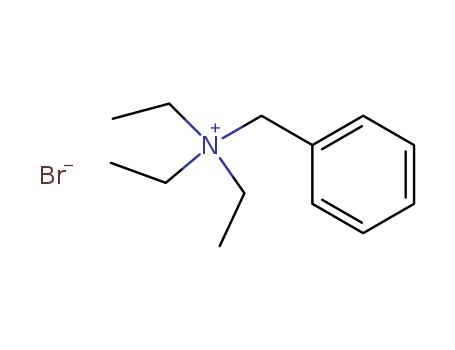171596-29-5
- Product Name:Tadalafil
- Molecular Formula:C22H19N3O4
- Purity:99%
- Molecular Weight:389.411
Product Details;
CasNo: 171596-29-5
Molecular Formula: C22H19N3O4
Appearance: white to off-white cyrstalline solid
Good Price Top Quality of Tadalafil 171596-29-5 In Medicine
- Molecular Formula:C22H19N3O4
- Molecular Weight:389.411
- Appearance/Colour:white to off-white cyrstalline solid
- Vapor Pressure:5.29E-13mmHg at 25°C
- Melting Point:298-300 °C
- Refractive Index:1.705
- Boiling Point:679.1 °C at 760 mmHg
- PKA:16.68±0.40(Predicted)
- Flash Point:364.5 °C
- PSA:74.87000
- Density:1.51 g/cm3
- LogP:2.08710
Tadalafil(Cas 171596-29-5) Usage
|
Indications and uses |
Cialis (or Tadalafil) was developed by American pharmaceutical company Lilly. It is used to treat erectile dysfunction and belongs to a second generation of PDE5 inhibitors. Studies show that Cialis works very quickly, taking effect in around 15-20 minutes, and has a prolonged effect that can last for up to 36 hours. T1/2 is 17.5h. |
|
Clinical Research |
In a study of 348 cases of mild to severe erectile dysfunction, patients were randomly given 20mg of Cialis or a placebo. Results showed that in comparison to the placebo group, patients who took Cialis experienced improved intercourse success in the 24-36 hours following medication, with many patients achieving successful sexual intercourse twice in 36 hours. Side effect rate and severity were also no different from those of the placebo group. Over 5% of patients in the Cialis group experienced headaches and indigestion. |
|
Side effects |
No severe negative reactions. No perceived facial flushing or sight abnormalities following medication. Rare cases of headache and indigestion. |
|
Warnings and precautions |
Patients currently taking nitrates, experiencing angina pectoris, suffering from heart disease, patients who have unregulated hypertension or hypotension, or patients who have had a stroke in the past 6 months should not take Cialis. |
|
Description |
Tadalafil (market name “Cialis” or “Adcirca”) is a kind of PDE5 inhibitor used for the treatment of erectile dysfunction, benign prostatic hypertrophy and pulmonary arterial hypertension. The effect of Tadalafil is relaxing the blood vessels muscles and increasing the blood flow into the corpus cavernosum. The mechanism of action of tadalafil is through inhibiting the activity of the cGMP specific phosphodiesterase type 5 (PDE5). PDE5 degrades cGMP in the corpus cavernosum located around the penis. Therefore, tadalafi leads to the increased concentration of cGMP which further causes the smooth muscle relaxation and increased blood flow into the corpus cavernosum. Some clinical studies also implied that tadalafil could improve endothelia function in men with increased cardiovascular risk and lower the urinary tract symptoms secondary to benign prostatic hyperplasia. |
|
Chemical Properties |
White to Off-White Cyrstalline Solid |
|
Originator |
Lilly/ICOS (US) |
|
Uses |
analgesic, norepinephrine uptake blocker, mu-opiod receptor agonist.A phosphodiesterase 5 inhibitor.Tadalafil is used for the treatment of erectile dysfunction. |
|
Definition |
ChEBI: A pyrazinopyridoindole that is 2,3,6,7,12,12a-hexahydropyrazino[1',2':1,6]pyrido[3,4-b]indole-1,4-dione substituted at position 2 by a methyl group and at position 6 by a 1,3-benzodioxol-5-yl group (the 6R,12aR |
|
Brand name |
Cialis (Lilly). |
|
General Description |
Tadalafil, (6R-trans)-6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2-methyl-pyrazino[1', 2' :1,6]pyrido[3,4-b]indole-1,4-dione (Cialis), is a potent PDE5 inhibitor.It received FDA approval for the treatment of erectiledysfunction in December 2003. Because of its half-life of17.5 hours, it is marketed as a 36-hour treatment. Tadalafil ispredominantly metabolized by hepatic enzymes, includingCYP3A4. The concomitant use of CYP3A4 inhibitors suchas ritonavir, indinavir, ketoconazole, as well as moderateCYP3A inhibitors such as erythromycin have been shown toresult in significant increases in tadalafil plasma levels.Much like sildenafil, tadalafil is under clinical investigationfor managing PAH. |
|
Mechanism of action |
Tadalafil was the last agent to be released and can be taken on a full stomach without slowing the onset. It has a much longer duration of action, lasting up to 48 hours, compared with sildenafil and vardenafil, which last for approximately 4 hours. The longer half-life of tadalafil results in a lengthened period of responsiveness as compared to sildenafil and vardenafil. This longer therapeutic window requires fewer time constraints for the effectiveness of tadalafil and has been interpreted as being advantageous through providing the option for more spontaneous sexual activity. Because of its long half-life, however, tadalafil, has been detected in plasma even 5 days after oral administration. This suggests the possibility of accumulation if taken regularly and in short intervals, which may result in an increased risk of side effects with the excessive use of this PDE5 inhibitor. The 3,4-methylenedioxy substitution on the phenyl ring was significant for increasing its potency as PDE5 inhibitor. Optimization of the chain on the piperazinedione ring resulted in no significant change in IC50s. Tadalafil is a highly potent PDE5 inhibitor (IC50, 5 nM), with high selectivity for PDE5 versus PDE1 through PDE4. The PDE5/PDE6 selectivity ratio is 85. |
|
Pharmacokinetics |
Tadalafil is different in structure from both sildenafil and vardenafil. It is rapidly absorbed and peaks in concentration (378 μg/L after a 20-mg dose) after 2 hours, displaying a long half-life of 17.5 hours. It also is metabolized by the liver (CYP3A4). Notably, its pharmacokinetics is not clinically influenced by alcohol or food intake or by factors such as diabetes or impaired hepatic or renal function. |
|
Clinical Use |
Tadalafil is one of the two new PDE5 inhibitors launched for the oral treatment of male erectile dysfunction. Tadalafil is a b-carboline derivative and it is structurally distinct from vardenafil (Levitraw) and sildenafil (Viagraw), both of which are PDE5 inhibitors based on a fused pyrimidine core structure. Tadalafil is synthesized in three steps starting from D-tryptophan methyl ester, by first condensing with piperonal in a Pictet-Spengler cyclization reaction to form the tetrahydro-β-carboline derivative, which is followed by chloroacetylation of the piperidine ring nitrogen and cyclization with methylamine. Tadalafil is a potent and highly selective inhibitor of PDE5 (IC50=1 nm). It shows >10,000-fold selectivity for PDE5 versus PDE1, 2, 3, 4, 7, 8 and 9, and >700-fold selectivity versus PDE6. Typically administered at 10 and 20 mg doses, tadalafil is rapidly absorbed and has a tmax of 2 h, which is slightly longer than those of sildenafil (1 h) and vardenafil (0.75 h). Clinically, all of these agents appear to have efficacy for many men within 30–60 min. However, tadalafil distinguishes itself from other PDE5 inhibitors in terms of significantly longer duration of action. The half-life of tadalafil dosed at 20 mg is 17.5 h as compared with 3.8 h for sildenafil (100 mg) and 4.7 h for vardenafil (20 mg). In clinical studies, significant rates of response were reported up to 36 h following drug ingestion. Tadalafil is predominantly metabolized in the liver by CYP3A4 to entities that are not active against PDE5 and excreted mainly as metabolites in the feces and the urine. The pharmacokinetics of tadalafil are unaffected by factors such as intake of food and alcohol, age, the presence of diabetes, and mild or moderate hepatic insufficiency. The most common drug-related adverse events are headache, back pain, dyspepsia, and myalgia. At 10 and 20 mg doses, Tadalafil does not have a significant effect on blood pressure and heart rate and does not result in increased instances of myocardial infarction. Rare reports of prolonged erections greater than 4 h and priapism have been noted with the use of tadalafil. Priapism, if not treated properly, can result in irreversible damage to the erectile tissue. Patients who have an erection lasting greater than 4 h are advised to seek emergency medical attention. Tadalafil has a modest synergistic effect on the nitrate-induced reduction in blood pressure and, as with sildenafil and vardenafil, it is contraindicated for use in patients on nitrate therapy. In diabetic patients, improvement of erectile function by tadalafil is irrespective of the type of diabetes and the type of diabetic therapy. |
|
Synthesis |
D-(-)-Tryptophan methyl ester (175) and 1,3- benzodioxole-5-carboxaldehyde (176) were subjected to a modified Pictet-Spengler reaction to form cis- and transtetrahydro- β-carboline tricyclic compounds. The ciscompound 177 was isolated as a white solid in 42% yield. The basic nitrogen in the piperidine ring of 177 was acylated with chloroacetyl chloride (179) to give compound 180 in 93% yield. Finally, the diketonepiperazine ring was formed by adding 180 to 33% methylamine in ethanol under refluxing conditions and yielded tadalafil (XXII) in 77% as a white solid. |
|
Drug interactions |
Potentially hazardous interactions with other drugs Alpha-blockers: enhanced hypotensive effect - avoid concomitant use. Antibacterials: concentration possibly increased by clarithromycin and erythromycin; concentration reduced by rifampicin - avoid. Antifungals: concentration increased by ketoconazole - avoid; concentration possibly increased by itraconazole. Antivirals: concentration possibly increased by fosamprenavir and indinavir; increased by ritonavir - avoid; increased risk of ventricular arrhythmias with saquinavir - avoid; avoid high doses of tadalafil with telaprevir. Cobicistat: concentration of tadalafil possibly increased - reduce dose of tadalafil. Nicorandil: possibly enhanced hypotensive effect - avoid concomitant use. Nitrates: enhanced hypotensive effect - avoid concomitant use. Riociguat: enhanced hypotensive effect - avoid concomitant use. |
|
Metabolism |
Tadalafil is metabolised in the liver mainly by the cytochrome P450 isoenzyme CYP3A4. The major metabolite, the methylcatechol glucuronide, is inactive. Tadalafil is excreted, mainly as metabolites, in the faeces (61% of the dose), and to a lesser extent the urine (36% of the dose). |
InChI:InChI=1/C22H23N3O3/c1-24-8-9-25-17(22(24)26)11-15-14-4-2-3-5-16(14)23-20(15)21(25)13-6-7-18-19(10-13)28-12-27-18/h2-7,10,15,17,20-21,23H,8-9,11-12H2,1H3/t15?,17-,20?,21-/m1/s1
171596-29-5 Relevant articles
Preparation method of tadalafil
-
Paragraph 0052-0054; 0056; 0059; 0061; 0064; 0066, (2022/01/12)
The invention provides a preparation met...
Preparation method of tadalafil
-
Paragraph 0015; 0041-0042, (2022/03/27)
The invention discloses a preparation me...
Preparation method of phosphodiesterase inhibitor
-
Paragraph 0016; 0056-0070, (2021/11/03)
The invention discloses a preparation me...
TADALAFIL SYNTHESIS METHOD
-
Page/Page column 11-22, (2020/11/03)
?The invention relates to a new one-pot ...
171596-29-5 Process route
-
![(6R,12aR)-6-(3,4-dihydroxyphenyl)-2-methyl-2,3,6,7,12,12a-hexahydropyrazino[1',2'-1,6]-pyrido[3,4-b]indole-1,4-dione](/upload/2023/8/346a5e0c-02bb-440c-8083-938df94cb1d5.png)
- 171489-03-5
(6R,12aR)-6-(3,4-dihydroxyphenyl)-2-methyl-2,3,6,7,12,12a-hexahydropyrazino[1',2'-1,6]-pyrido[3,4-b]indole-1,4-dione

-
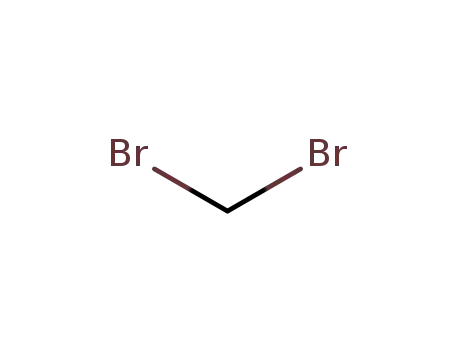
- 74-95-3
1,1-dibromomethane

-
![(6R,12aR)-2,3,6,7,12,12a-hexahydro-2-methyl-6-(3,4-methylenedioxyphenyl)-pyrazino[2',1':6,1]pyrido[3,4-b]indole-1,4-dione](/upload/2023/8/e89e7542-da34-45a4-b473-f1408dd46eb5.png)
- 171596-29-5
(6R,12aR)-2,3,6,7,12,12a-hexahydro-2-methyl-6-(3,4-methylenedioxyphenyl)-pyrazino[2',1':6,1]pyrido[3,4-b]indole-1,4-dione
| Conditions | Yield |
|---|---|
|
With sodium hydroxide; In N,N-dimethyl-formamide; at 60 ℃; for 6h;
|
95% |
|
With caesium carbonate; In N,N-dimethyl-formamide; at 80 ℃; for 8h;
|
93% |
|
With caesium carbonate; In N,N-dimethyl-formamide; at 80 ℃; for 8h;
|
93% |
|
With caesium carbonate; In N,N-dimethyl-formamide; at 80 ℃; for 8h;
|
93% |
-
![(1R,3R)-methyl-1,2,3,4-tetrahydro-2-(2-(benzyl(methyl)amino)acetyl)-1-(3,4-methylenedioxyphenyl)-9H-pyrido[3,4-b]indole-3-carboxylate](/upload/2023/8/9033c471-6c71-46af-b628-74ed998d4bf8.png)
- 1224724-00-8
(1R,3R)-methyl-1,2,3,4-tetrahydro-2-(2-(benzyl(methyl)amino)acetyl)-1-(3,4-methylenedioxyphenyl)-9H-pyrido[3,4-b]indole-3-carboxylate

-
![(6R,12aR)-2,3,6,7,12,12a-hexahydro-2-methyl-6-(3,4-methylenedioxyphenyl)-pyrazino[2',1':6,1]pyrido[3,4-b]indole-1,4-dione](/upload/2023/8/e89e7542-da34-45a4-b473-f1408dd46eb5.png)
- 171596-29-5
(6R,12aR)-2,3,6,7,12,12a-hexahydro-2-methyl-6-(3,4-methylenedioxyphenyl)-pyrazino[2',1':6,1]pyrido[3,4-b]indole-1,4-dione
| Conditions | Yield |
|---|---|
|
With hydrogen; Raney Ni; In ISOPROPYLAMIDE; at 80 ℃; for 22h; under 2280.15 Torr; Product distribution / selectivity;
|
94% |
171596-29-5 Upstream products
-
74-89-5
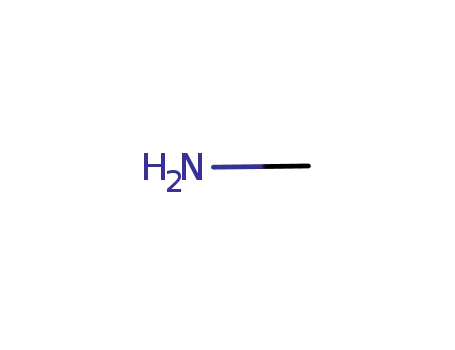
methylamine
-
171489-59-1
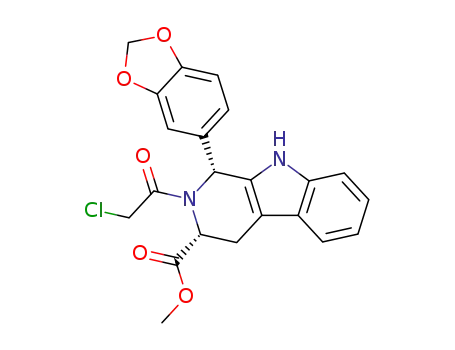
methyl (1R,3R)-1-(3,4-methylenedioxyphenyl)-2-chloroacetyl-2,3,4,9-tetrahydro-9H-pyrido[3,4-b]indol-3-carboxylate
-
749864-18-4
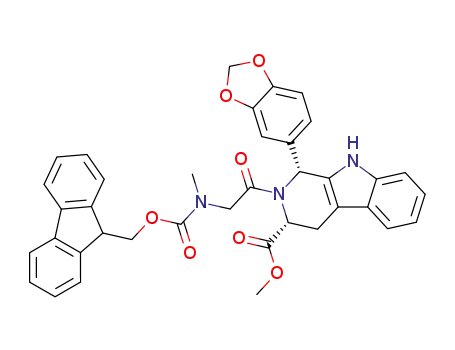
1-benzo[1,3]dioxol-5-yl-2-{[(9H-fluoren-9-ylmethoxycarbonyl)-methyl-amino]-acetyl}-2,3,4,9-tetrahydro-1H-β-carboline-3-carboxylic acid methyl ester
-
951661-81-7

(1R,3R)-1-(benzo[d][1,3]dioxol-5-yl)-2-(2-chloroacetyl)-N-methyl-2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indole-3-carboxamide
171596-29-5 Downstream products
-
531500-48-8
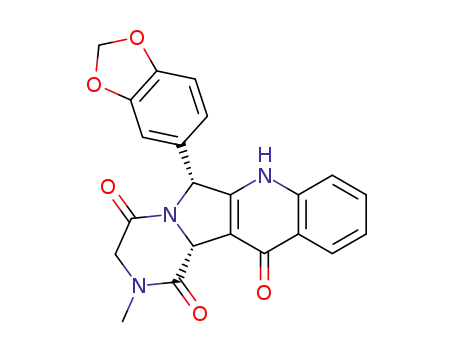
11-benzo[1,3]dioxol-5-yl-3-methyl-2,3,4a,11-tetrahydro-10H-3,10,11a-triaza-benzo[b]fluorene-1,4,5-trione
-
171596-27-3
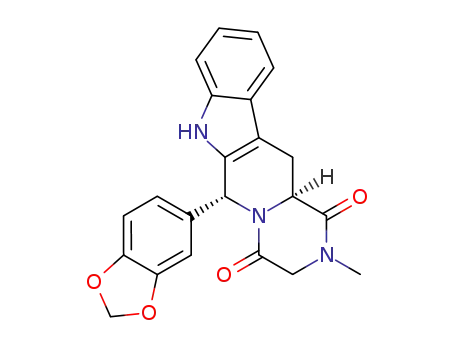
(6R,12aS)-2,3,6,7,12,12a-hexahydro-2-methyl-6-(3,4-methylenedioxyphenyl)-pyrazino[2',1':6,1]pyrido[3,4-b]indole-1,4-dione
-
1220393-12-3
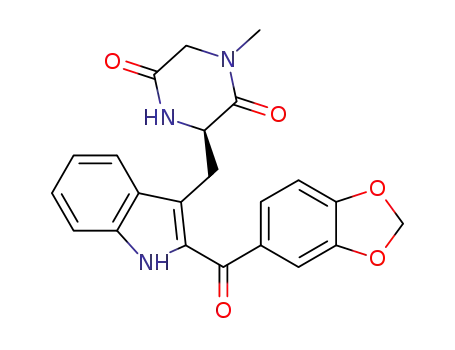
3-((2-(benzo[d][1,3]dioxole-5-carbonyl)-1H-indol-3-yl)methyl)-1-methylpiperazine-2,5-dione
-
1242099-13-3

C10H12O3*C22H19N3O4
Relevant Products
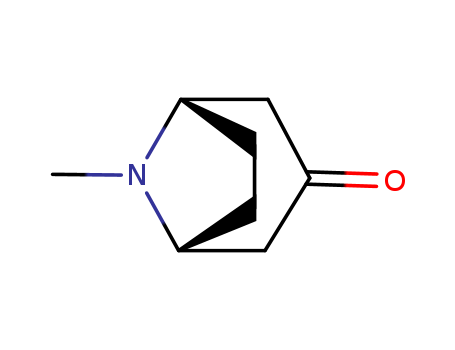
-
Tropinone
CAS:532-24-1
-
Benzyltriethylammonium bromide
CAS:5197-95-5
-
Anastrozole
CAS:120511-73-1